Cryo-electron tomography (cryo-ET) offers unprecedented, high-resolution insights into the molecular ultrastructure of cells. However, locating specific targets and preparing the perfect electron-transparent lamella remains a significant bottleneck. Traditionally, researchers had to rely on a stand-alone fluorescence microscope (FM) to identify targets before physically transferring the delicate grid to the FIB-SEM. This transfer is inherently risky, prone to ice contamination, and makes precise correlation difficult.
The introduction of integrated fluorescence microscopes (iFMs) greatly reduced these risks by housing the FM directly inside the FIB-SEM chamber. Yet, in most standard iFM setups, correlation and stage movements are still required to switch between optical imaging and FIB milling modes. Today, there are a few advanced options out there that attempt to eliminate these stage moves entirely. The ultimate goal for maximum precision is a tri-coincident system where the light, electron, and ion beams intersect at the same focal point, enabling real-time monitoring without moving the sample.

Figure 1. General concept of the tri-conincidence imaging
Comparing tri-coincident designs
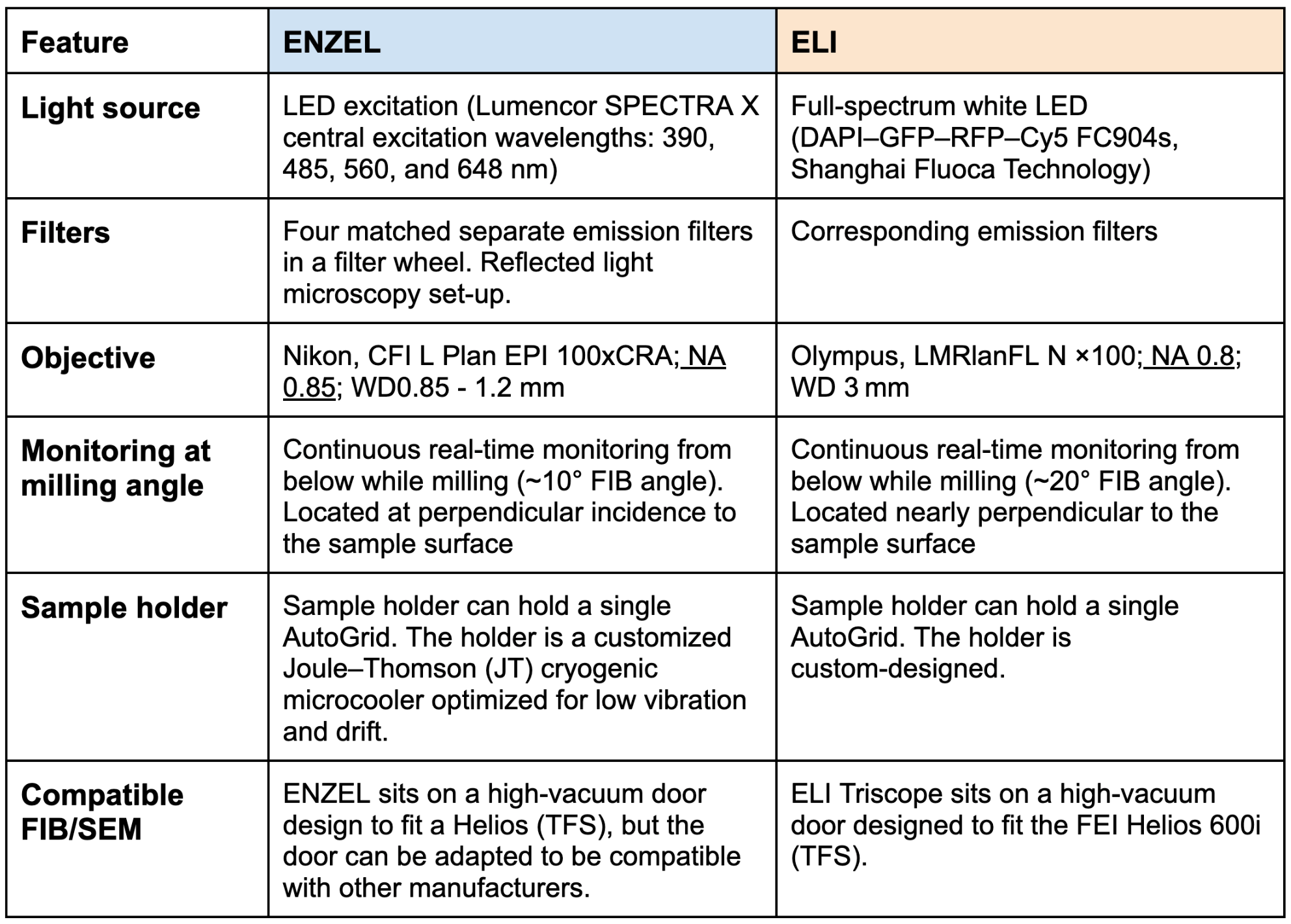
When evaluating tri-coincident setups such as our own Delmic ENZEL system and the ELI TriScope system, several key design choices determine how seamlessly the workflow is. The TFS Arctis is not considered in this specific "tri-coincident targeting" discussion because a mechanical stage rotation is still necessary between the optical and FIB-SEM imaging positions.

Figure 2. Set-ups of ENZEL and ELI TriScope. Both setups use a similar design in which the objective lens (OL) is positioned beneath the sample to facilitate tri-coincidence imaging.
Images are adapted from: ENZEL: Boltje et al., eLife, 2022;11:e82891. ELI TriScope: Li et al., Nat Meths. 2023;20(2):276–283
Here is a brief comparison of the two tri-coincident setup designs:
Ways of targeting: A look at recent literature
Tri-coincident microscopy fundamentally changes how we approach lamella preparation by enabling new targeting strategies. Here is how three recent papers utilize these systems to pioneer distinct, highly effective ways of targeting:
1. Targeting via live fluorescence monitoring
Boltje et al. (eLife, 2022) and Li et al. (Nat. Methods, 2023) revolutionized targeting by using live, multi-color fluorescence monitoring on the ENZEL and ELI-Triscope, respectively. By tracking the fluorescence signal in real-time throughout the milling process, researchers can avoid time-consuming sample repositioning and verify target integrity based on fluorescence visualization and monitoring of the fluorescent intensity. By relying on this direct method, the team of Wang (bioRxiv, 2024) successfully stopped milling at the right depth to capture dense inflammasome condensates at various stages, achieving an impressive target retention rate (52 out of 82 lamellae) for final cryo-ET imaging.
Video 1: Adapted from Boltje et al., eLife, 2022;11:e82891
2. Targeting via fluorescence-based optical interference
Sica et al. (Nature Communications, 2026) demonstrated that the milling process itself functions as a powerful optical tool, driven by predictable oscillations in fluorescence intensity. As the lamella reaches nanometer-scale thickness, it acts as an optical cavity, causing the fluorescent signal to oscillate. These oscillations function as an "optical ruler", where each peak and valley corresponds to a measurable decrease in thickness. By tracking these cycles, you can precisely determine your depth and stop milling the moment the signal drops, confirming the target has been reached. This real-time monitoring allows operators to track the target with a precision that goes beyond the diffraction limit of light. This allows you to reliably capture rare, small structures, like localized virions inside host cells, without relying on fluorescent fiducials or complex axial registration.

Figure 3. This graph illustrates the normalized fluorescence intensity of beads as milling progresses (Blue line). By tracking the interference patterns, the team can pinpoint the bead's exact Z-position within the ice, ensuring the final lamella contains the target of interest.
3. Targeting with multimodal real-time feedback
Boltje et al. (Cell Reports Methods, 2025) took a comprehensive approach, showing that successful targeting isn't just about locating the structure, but also ensuring the lamella itself is perfectly crafted. To achieve this, they developed three distinct strategies to monitor lamella quality in real time (Figure 4). First, they determine lamella thickness using 4D-STEM, where electron scattering patterns are recorded using a scintillator combined with ENZEL's optical system. Second, they utilize reflected light microscopy (RLM) to estimate thickness and assess the protective platinum (Pt) layer. By recording a z-stack to bring both the top and bottom of the lamella into focus, operators can measure thickness from the foil side and visually check the uniformity of the Pt layer. Finally, they leverage thin-film interference to precisely measure and generate a comprehensive 2D thickness map of the entire lamella.

Figure 4. Image adapted from Boltje et al. Cell Reports Methods, 2025
Looking Ahead
While tri-coincident systems already represent a massive leap forward in lamella success rates, the journey doesn't end here. Current workflows remain largely manual, and the full potential of High-Pressure Freezing (HPF) samples in these setups is waiting to be unlocked. At Delmic, we envision an automated coincident milling process that eliminates guesswork, ensuring consistent lamella thickness and flawless target retention every single time. Furthermore, as the cryo-ET field increasingly turns its attention toward the structural mysteries of bulkier, complex tissues, we see the integration of HPF as a critical next step in this evolution.
Conclusion
The horizon of tri-coincident microscopy is incredibly exciting and promises to push structural biology into new frontiers. However, it is worth remembering that non-coincident integrated setups, like our METEOR system, remain highly robust, accessible, and proven solutions for many of today's cryo-ET workflows. By eliminating a risky transfer step, METEOR dramatically reduces ice contamination and streamlines correlation, making it an incredibly robust choice for laboratories worldwide.
We are constantly innovating and refining our technologies, so stay at the forefront of these developments by signing up for our newsletter and following us on LinkedIn.
.png)




