Achieving the proper balance between luminescence and high contrast to simultaneously study specific proteins but also to acquire ultrastructural information is one of the challenges of bioimaging. CLEM is an imaging technique for optoelectronic investigation of structure-function relationships at the nanoscale, which has gained importance in recent years. However, the strong mismatch between optical and electron imaging resolutions is still a limiting factor for CLEM.
Advantages of correlative cathodoluminescence electron microscopy
One technique that could be an attractive alternative is CCLEM. This bioimaging technique can be successfully used for studying the structure and composition of cells and tissues: with cathodoluminescence generated by an electron beam, it is possible to obtain luminescence data with a much higher spatial resolution than is attainable in a conventional optical microscope.
CCLEM allows to not only overcome a mismatch between optical and electron imaging resolutions, it also opens new possibilities for using luminescent labels. Commonly, organic fluorophores are used for labelling the biological specimen. However, with CCLEM technique inorganic materials, which emit photons upon electron beam exposure and are more stable, can be a good alternative.
Available luminescent labels
CCLEM is currently studied and applied by a research group at EMPA in Switzerland. The group has already published three papers [1,2,3] on CCLEM imaging, which investigate the performance of various labels for this bioimging. Since organic proteins and dyes are rather unstable under electron irradiation and produce low CL emission, the authors of the papers investigate various exogenous luminescent labels and their feasibility for photoluminescence and cathodoluminescence bioimaging setting. One of the two papers [2] concluded that compared to organic fluorophores and semiconductor quantum dots, two types of RE3+ doped nanocrystals (Y2O3:Tb3+ and YVO4:Bi3+,Eu3+) showed the best results in terms of emission stability and brightness.
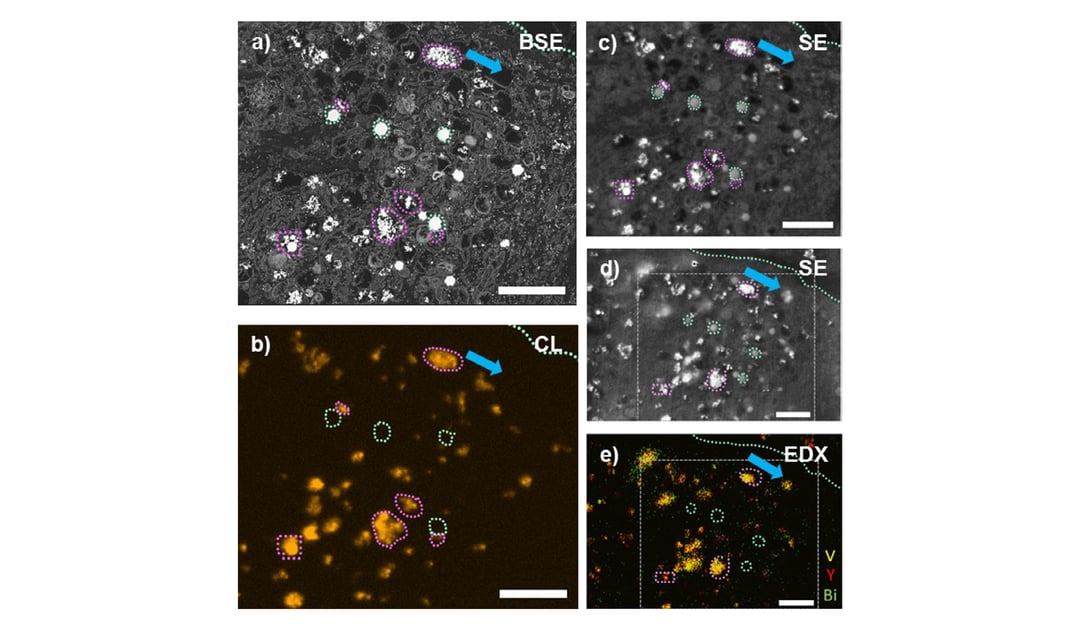
The most recent paper from EMPA [3] demonstrates that specific molecules can be visualized at nanoscale resolution in the context of the cellular ultrastructure with CCLEM-based immunolabeling (immunoCCLEM). This paper presents for the first time the utility of rare-earth element doped inorganic nanoparticle-labels. Characteristic receptor clustering on the surface of cancer cells was identified by specific labelling of the receptors with folic acid decorated nanoparticles showing single particle CL emission. This study showcases the feasibility of immunoCCLEM for protein localization in the context of the cellular ultrastructure in EM.
EMPA’s studies show that CCLEM can be a powerful tool for gaining insights into the interaction of engineered nanoparticles with cells, which could be potentially applied in pathophysiological research and molecular biology.
To learn more about CCLEM as a bioimaging technique, we suggest you watch this webinar, where our applications specialist Sangeetha Hari talks about cathodoluminescence probes and exciting applications of CCLEM.
References
[1] Ultrabright and Stable Luminescent Labels for Correlative Cathodoluminescence Electron Microscopy Bioimaging Keevend, L. Puust, K. Kurvits, L. R. H. Gerken, F. H. L. Starsich, J.-H. Li, M. T. Matter, A. Spyrogianni, G. A. Sotiriou, M. Stiefel, I. K. Herrmann, Nano Lett. (2019)
[2] Tb3+-doped LaF3nanocrystals for correlative cathodoluminescence electron microscopy imaging with nanometric resolution in focus ion beam-sectioned biological samples Keevend, M. Stiefel, A. L. Neuer, M. T. Matter, A. Neels, S. Bertazzo, and I. K. Herrmann. Nanoscale 9, 4383-4387 (2017)
[3] Correlative Cathodoluminescence Electron Microscopy: Immunolabeling Using Rare‐Earth Element Doped Nanoparticles Keevend, R. Krummenacher, E. Kungas, L. R. H. Gerken, A. Gogos, M. Stiefel, and I. K. Herrmann. Small 16 (2020)
.png)