One of the perovskite-based devices attracting the attention of scientists and energy companies is the perovskite solar cells. Recent researches demonstrate that perovskite solar cells have shown a significant increase in power conversion efficiencies (the percentage of sunlight converted to electricity) from 3.8% in 2009 to 22.1% in 2016 [1] and to 28% in 2018 [3]. If the efficiency can be increased, even more, the perovskite solar cells can potentially become a cheaper and more flexible source of solar energy.
Researchers and electronic designers are looking for ways to improve the performance of perovskite materials. Quite a few challenges in the production and quality of perovskite solar cells separate them from being widely commercialized. For example, perovskite solar cells still can’t demonstrate the same stability as silicon, which is a common material used for solar panels production [3].
Understanding various perovskite materials and their efficiency is the key to the successful development of stable solar cells. Various combinations and possibilities in the perovskite structure lead to multiple challenges. One of such examples is CsPbIBr2, a fully inorganic perovskite material in which a mix of iodide and bromide is present [1]. Using a mixed halide composition, it is possible to boost power conversion efficiencies. However, it is known that under the influence of external stimuli such as light the I and Br halogen ions can move within the perovskite material leading to phase segregation. Such segregation can have a major impact on the electrical properties of the material and its function as a solar cell or light-emitting device [4,5].
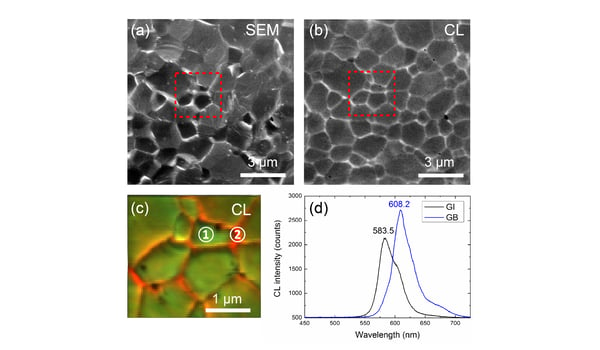
A great tool to understand phase segregation is cathodoluminescence (CL) imaging. Using hyperspectral imaging, it was possible to visualize the changes in the emission spectrum and conclude that the grain boundaries are richer in iodine than the grain interiors, leading to brighter emission and a redshifted emission spectrum. Read the full application note on phase segregation in inorganic CsPbIBr2.
The figure below shows a panchromatic CL intensity map revealing light emission efficiency at different points on the material. False-color CL image derived from a hyperspectral CL scan visualizing differences in emission spectrum from point to point. Experiments were performed on the SPARC system (Australian Research Council grant LE140100104) at the Monash Centre for Electron Microscopy (MCEM, Monash University, Australia).
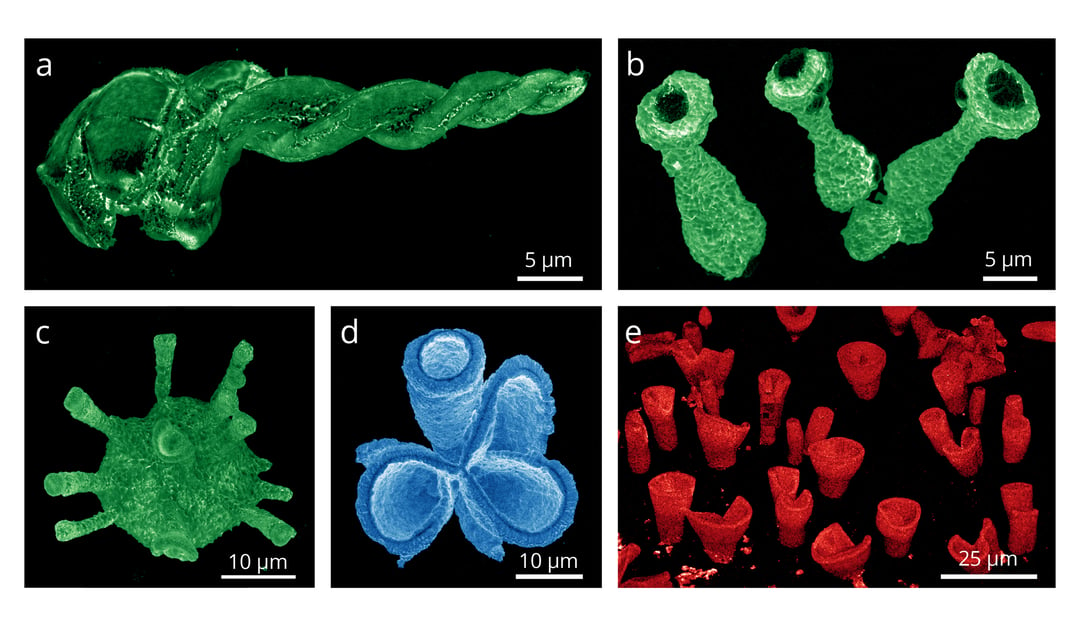
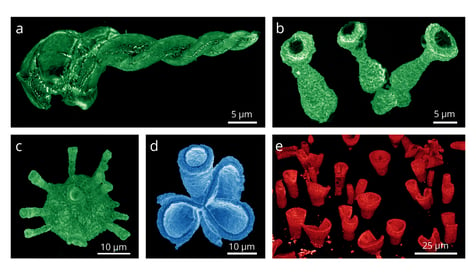
 Another type of perovskite material, methylammonium lead halide perovskite, which have gained significant interest due to their exceptional performance in solar cell, can also be studied with the cathodoluminescence technique. It was used to study unconventional 3D shapes of perovskites, which were grown using carbonate salts. By obtaining cathodoluminescence intensity maps, it was proven that there is (efficient) light emission throughout the structures (read more about it in the application note). The image below demonstrates the cathodoluminescence (CL) intensity maps of synthetically grown 3D perovskite structures acquired with the SPARC CL system (credits: Noorduin Lab, AMOLF).
Another type of perovskite material, methylammonium lead halide perovskite, which have gained significant interest due to their exceptional performance in solar cell, can also be studied with the cathodoluminescence technique. It was used to study unconventional 3D shapes of perovskites, which were grown using carbonate salts. By obtaining cathodoluminescence intensity maps, it was proven that there is (efficient) light emission throughout the structures (read more about it in the application note). The image below demonstrates the cathodoluminescence (CL) intensity maps of synthetically grown 3D perovskite structures acquired with the SPARC CL system (credits: Noorduin Lab, AMOLF).
Being able to visualize this phenomenon at these small length scales is critical for understanding perovskite materials. This knowledge can be used to harness positive effects to improve device performance.
References
[1] W. Li et al. Phase Segregation Enhanced Ion Movement in Efficient Inorganic CsPbIBr2 Solar Cells Adv. Energy Mater. 7, 1700946 (2017)
[2] K. Stacey. A better method for making perovskite solar cells. News from Brown. Retrieved from: https://news.brown.edu/articles/2015/03/perovskite (2015)
[3] J. Kumagai. Power From Commercial Perovskite Solar Cells Is Coming Soon. IEEE Spectrum. Retrieved from: https://spectrum.ieee.org/energy/renewables/power-from-commercial-perovskite-solar-cells-is-coming-soon (2019)
[4] D. J. Slotcavage et al. Light-Induced Phase Segregation in Halide-Perovskite Absorbers, ACS Energy Lett. 1, 1199-1205 (2016)
[5] Rachel E. Beal et al. Cesium Lead Halide Perovskites with Improved Stability for Tandem Solar Cells J. Phys. Chem. Lett. 7, 746-751 (2016)
.png)