This technique is becoming more and more important since knowing the location, as well as the structure of proteins, is crucial for understanding their cellular function. Cryo-ET holds a mountain of promise for furthering our understanding of all biological processes. However, the current sample preparation method is the major bottleneck in unleashing this technique. Curious to learn how to optimize the current cryo-ET workflow? Keep reading or watch the webinar.
The current Cryo-ET workflow
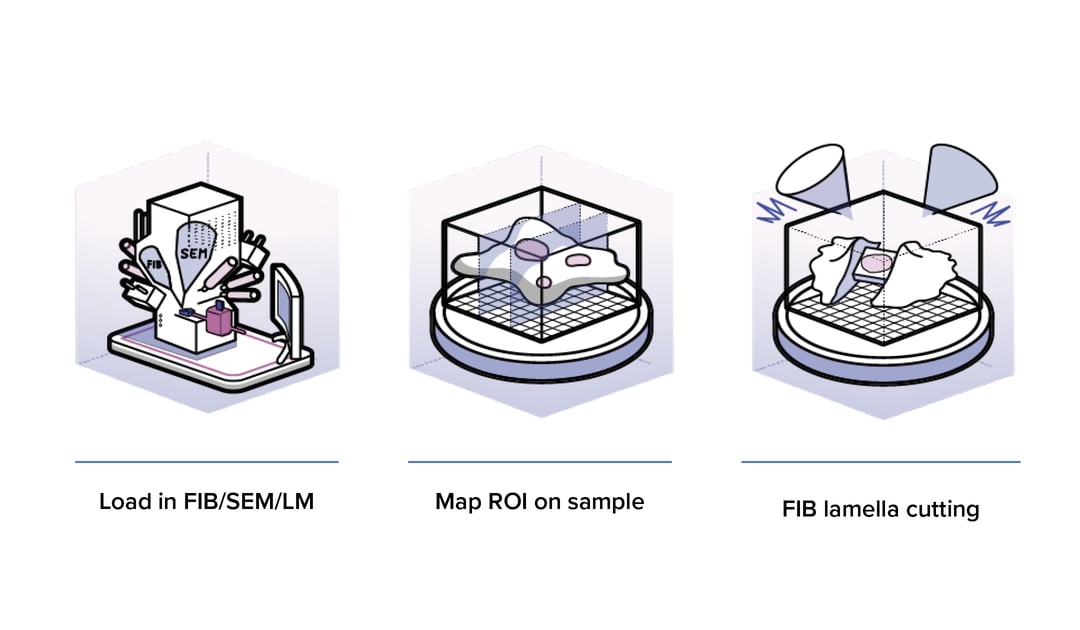
In cryo-ET a sample is flash frozen, thinned to the appropriate thickness (100-300 nm) and a series of images are captured with a transmission electron microscope (TEM). These 2D images are used to reconstruct a 3D image, called a tomogram. During all these steps the sample must remain in a vitrified state at ~ -160°C to avoid crystalline ice formation.
To create such a thin section the use of a Focused Ion Beam with a Scanning Electron Microscope (FIB/SEM) has become the gold standard. The FIB is used to mill away the surrounding material and create a thin section, called a cryo-lamella. Identifying the region of interest (ROI), in order to mill in the right location is a crucial step, since being off target for this process could result in milling away your structure of interest. To overcome this, cryo-Fluorescence Microscopy (cryo-FM) is often used to identify the ROI and avoid ‘blind’ milling.
In this technique, fluorescent markers are used to label the structures or proteins of interest, which are then found back with the SEM part of the FIB/SEM. However, the incorporation of a cryo-FM in the cryo-ET workflow brings about many challenges. Firstly, transfer of the sample to the cryo-FM before milling (to identify ROIs) significantly increases the handling of the sample and thereby increases the risk of contaminating or damaging the sample. Ideally, the sample is also inspected again with the cryo-FM after milling to ensure the ROI is not milled away. However, this is so risky that this is currently not done at all. Secondly, the correlation of the fluorescence image with the image in the FIB/SEM is not an easy task. It requires markers for navigating to the correct spot on the sample and if the overlay of the fluorescence and SEM images is not exactly correct, the ROI could still be missed or milled away.
Possibilities of integrated cryo-Fluorescence Microscopy
To overcome these challenges, Delmic is currently developing a cryo-FLM solution that can be integrated into a FIB/SEM system. Having a fluorescence microscope inside the FIB/SEM greatly reduces the amount of handling steps required, resulting in less contamination, less damage, and a more efficient cryo-ET workflow. Additionally, switching between FM and SEM imaging without moving the sample allows direct correlation of the SEM and FM images. This significantly speeds up the identification of ROIs and ensures the milling process is performed at the correct site and does not endanger the structure of interest. Furthermore, it is easy to reinspect the sample with FM after the milling process, thereby improving the quality of the samples that ultimately end up in the cryo-TEM.
Delmic's cryo-FM system is easily integrated in existing cryo-ET workflows and represents a major step forward in making the powerful cryo-ET technique substantially easier and significantly more efficient. Would you like to know more? Contact us directly for more information.
This work is supported by the European SME2 grant № 879673 - Cryo-SECOM Workflow
.png)